PT Prof. Joseph Roche and OT wife Renuka publish high-impact research on COVID-19 treatment possibility
In March, when the novel coronavirus was declared a pandemic and Wayne State University faculty began shifting their classes from in-person to online experiences, Eugene Applebaum College of Pharmacy and Health Sciences Assistant Professor of Physical Therapy Joseph A. Roche had t o deal with the ramp-down of onsite research even as he was busy poring over literature on COVID-19 and related infections.
o deal with the ramp-down of onsite research even as he was busy poring over literature on COVID-19 and related infections.
Vox covers Roches' COVID-19 research
"I believe that all scholars trained in the scientific method can contribute in some way in a crisis like this epidemic," he said. And he happened to be sheltering in place with someone who shares that conviction. His wife Renuka, who goes by Ray, is an assistant professor of occupational therapy at Eastern Michigan University who researches developmental neurology. "We went through the same PhD program at the University of Maryland, Baltimore, with a heavy biomedical science component."
Watch the Roches discuss their research
The two have collaborated on numerous papers since then, and they had a light-bulb moment for their next one while watching WDIV-TV Local 4 News coverage. "In a report on the evening of March 23, we heard Dr. Frank McGeorge talking about ACE inhibitors, ACE2 protein and COVID-19. This prompted us to work through the night and submit a response to WDIV Local 4, highlighting our bradykinin [BK] model for COVID-19 complications and the suggested use of the FDA-approved BK blocker icatibant," Roche said, adding with a chuckle, "The timestamp on our email to WDIV was 3-24-20 at 3:29 a.m."
McGeorge doesn't just play a doctor on TV; Local 4's health reporter is also a Henry Ford Hospital ER doctor, and the Roches thought he was on to something.
Since the disclosure of their BK model for COVID-19 complications to WDIV, the Roches have been relentless in their engagement with industry partners, funding agencies, publishers, the media, city/state/federal/international agencies, clinicians and researchers. It was out of these efforts that an Open Letter to the Scientific Community was published on the Figshare server on April 7. Even as their idea was under consideration by various entities, the Roches were working with the COVID-19 research leadership at Wayne State about taking their idea to clinical trials.
Anything that affects human movement is a PT's business. ... To live is to move and to move is to live.
Roche, who is a member of three professional bodies within the Federation of American Societies for Experimental Biology (FASEB), felt the need for the experimental biology community to have a primer on what is known and unknown about COVID-19. The Roches therefore prepared a more detailed description of their BK model for COVID-19 complications and explained the evolving knowledge of the symptomatology of COVID-19 from the lens of their model. This included a discussion on possible links between genetics and worse outcomes, and dysregulated BK signaling and the loss of smell and increased thirst. The Roches were careful to cite all original research literature that helped them develop their model and their suggestion for the use of icatibant. They also did due diligence in pointing out the limitations of their model. This work culminated in the submission of a Hypothesis article to FASEB's flagship journal.
The Roches submitted their article to the FASEB Journal on April 20. It was accepted just two days later on April 22. A week and a half after that, on May 2, "A Hypothesized Role for Dysregulated Bradykinin Signaling in COVID-19 Respiratory Complications" appeared on the FASEB website as the online version of record before inclusion in the June 2020 issue of the monthly journal.
A first decision from the FASEB Journal usually takes an average of three weeks. But these are not usual times. At the top of the Roches' paper is an editor's note saying that the article was fast-tracked under a recently instituted interim policy in which the editors may, at their discretion, accept coronavirus-related manuscripts without additional review.
"This is incredible news and we are so very, very proud," said Associate Dean for Health Sciences Peter Frade. "The FASEB Journal is highly regarded. This is just the beginning of continued success for Dr. Roche."
The paper begins: "As the COVIDâ€19 pandemic is rapidly spreading, and health delivery systems are being overwhelmed by the large numbers of patients needing acute care for breathing difficulty, it is imperative that safe and effective pharmacotherapeutic strategies are rapidly explored to improve survival. Since time is of the essence to reduce mortality in patients with COVIDâ€19 respiratory complications, repurposing FDAâ€approved drugs that have a good safety profile for offâ€label and/or compassionate use should be a strategic priority.
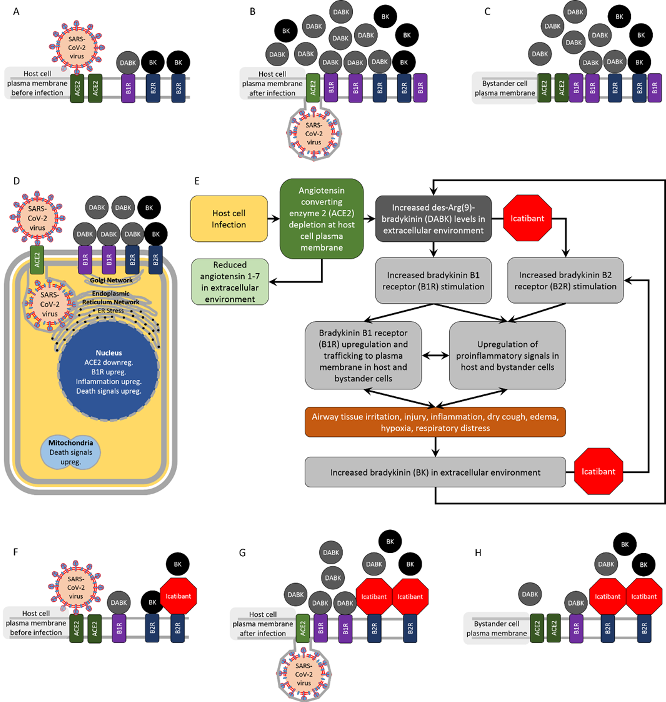
"Based on our examination of basic and clinical studies, we hypothesize that dysregulated bradykinin (BK) signaling is involved in COVIDâ€19 respiratory complications."
BK is a peptide that promotes inflammation. ACE inhibitors increase BK levels and are FDA approved for the treatment of hypertension and heart failure. The Roches postulated that the offâ€label use of icatibant, which is sold under the brand name Firazyr by Takeda Pharmaceuticals for the treatment of angioedema, seems promising for patients with unremitting respiratory distress caused by COVIDâ€19.
Angioedema is the rapid swelling of the area beneath the skin, normally an allergic reaction but it can also be hereditary. The swelling happens because fluid accumulates, and it tends to affect areas with loose areas of tissue, especially the face and throat.
"Icatibant is effective in treating breathing difficulty in patients presenting with angioedema, including angioedema caused by … ACE inhibitors taken for hypertension," the paper continues. "It might be purely coincidental that COVIDâ€19 causes a dry cough - a rare but characteristic side effect of ACE inhibitors, which is linked to BK."
In fact, Roche has a vivid childhood memory of his own mother coughing through the night for several weeks after being started on an ACE inhibitor, and uses this personal anecdote in his lectures on cardiovascular pharmacology.
Heartening validation
When FASEB Journal Associate Editor Charles N. Serhan sent the acceptance letter, he wrote, "We believe your hypothesis and potential role of the kinin system in COVID-19 is worth promoting. Your hypothesis is very interesting and hopefully, with publication in FJ, interest will emerge for a clinical trial as you've indicated."
Indeed, the paper has garnered a healthy amount of interest from researchers and medical professionals near and far. "The validation of our work is heartening," said Roche. Wayne State is engaged in discussions with industry partners on taking the next steps toward moving the BK blockade idea into the clinic. Roche also was invited by Dr. Joshua Zimmerberg, a National Institutes of Health intramural investigator, to give a virtual talk on his research on muscle and COVID-19 at the NIH on May 11.
"People say to me: You're a physical therapist - why is this any of your business?" Roche said. "Even my own brother, who's a GI physician, sometimes asks me why I have opinions on the pathophysiology and management of medical conditions that PTs don't directly treat.
"I tell him and everyone else: Anything that affects human movement is a PT's business. When movement is affected, it affects health, wellness and the quality of life. To live is to move and to move is to live."
It's no coincidence, then, that Roche's recent NIH talk was titled "Human Movement Is a Physical Therapist's Business." In it, he highlighted a local news story about the DMC Rehabilitation Institute of Michigan in which WSU alum Mark Mitchell, DPT '13 discusses helping a patient work through the movement consequences of COVID-19.
Roche trained in a molecular physiology lab for 12 years, has done pharmacology research and teaches pharmacology to PT students. "I feel like all my experience funneled together for this effort," he said. "I tapped into my background to put the idea together based on everything I've learned and know."
His co-author did the same. "When Ray worked as an acute care OT, she had patients whose respiratory complications required ventilation," Roche said. "That experience helped her better understand the clinical literature on COVID-19 and glean information pertinent to our bradykinin hypothesis."
All of the couple's research was supervised by another one of their collaborative efforts: 5-year-old Vanya, who watched her parents work with great interest during the early days of Michigan's Stay Home, Stay Safe executive order. "Our daughter saw us become maniacally obsessed with trying to use science to answer big questions," Roche said. "Now she's got it all planned out. She says that when we win the Nobel Prize, she'll spend the award money on toys and helping those in need."
Vanya learned about the prize after reading a book in school about President Barack Obama. "She thinks being awarded the Nobel Prize will be as simple as when Ray and I won the church talent show with our rendition of Maroon 5's Memories," Roche laughed.
"Even though she's seen her parents pounding our heads in frustration, she thinks it looks like fun," Roche said. "As we read the acceptance letter from the FASEB Journal, Ray, Vanya, and I hugged and cried happy tears, because we believed that this was one step closer toward lives being saved."
It's no wonder that despite the fact that Roche is pointing his kindergartner toward a career in surgery, Vanya has other plans: "She wants to be a scientist."
Roche, who believes his recently departed father is watching down on his work with pride, said that he would like to dedicate his COVID-19 research efforts to health care workers and scientists who are putting their lives at risk while trying to save others.